Dosing
Flexible dosing for those moments that demand Qlosi-clear near vision
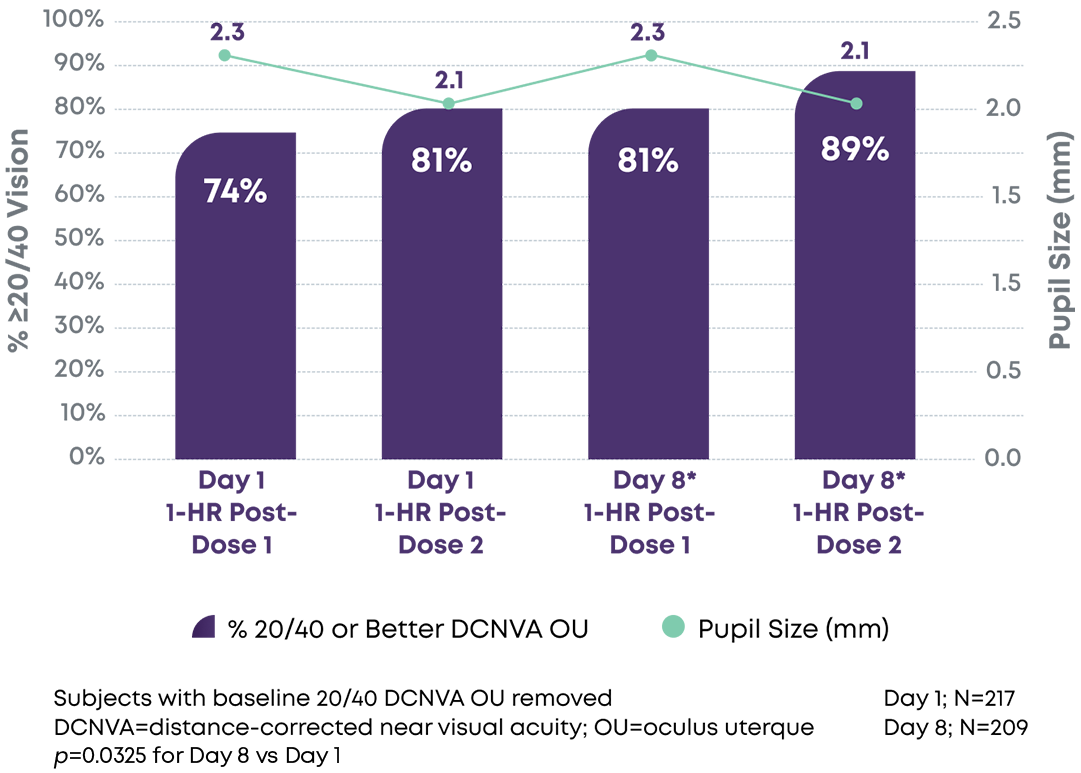
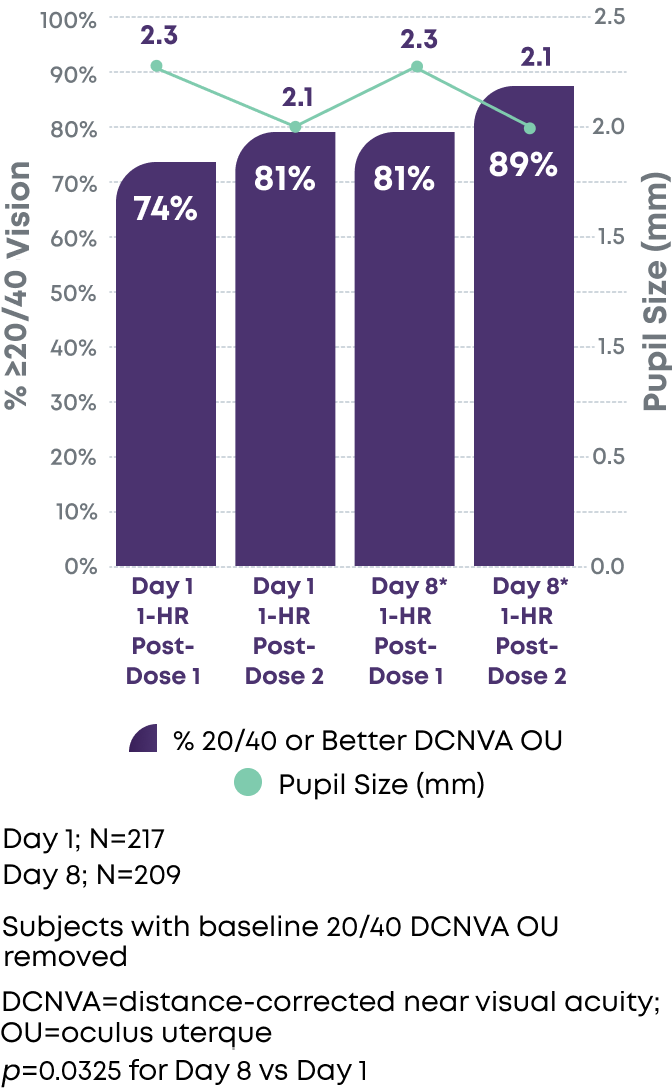
Consistently reduced pupil size with significant increase in near vision acuity responders likely due to neuroadaptation1-3
DCNVA 20/40 or better (N=206)
Pupil Size vs. % Achieving 20/40 Vision or Better DCNVA OU
Patients can choose 1 drop or 2, putting them back in control of their near vision4
Patients can use one drop of Qlosi in each eye for up to 3 hours of clear near vision

This dose can be repeated a second time after 2 to 3 hours for an effect up to 8 hours

The Qlosi Clear Start Week |
Help your patients achieve their near vision potential4
The Clear Start Week is for patients new to Qlosi and will help them adjust to their near vision. During this week, patients should be instructed to use Qlosi twice a day, every day. Consistent use at the start means the potential for better results over time as neuroadaptation takes effect.1-4
After the Clear Start week |
Patients can now take advantage of the flexible dosing based on individual needs and can use Qlosi up to 2 times a day.4*
*
Presbyopia patients start with 1 drop of Qlosi in each eye twice a day for 7 days in a row.